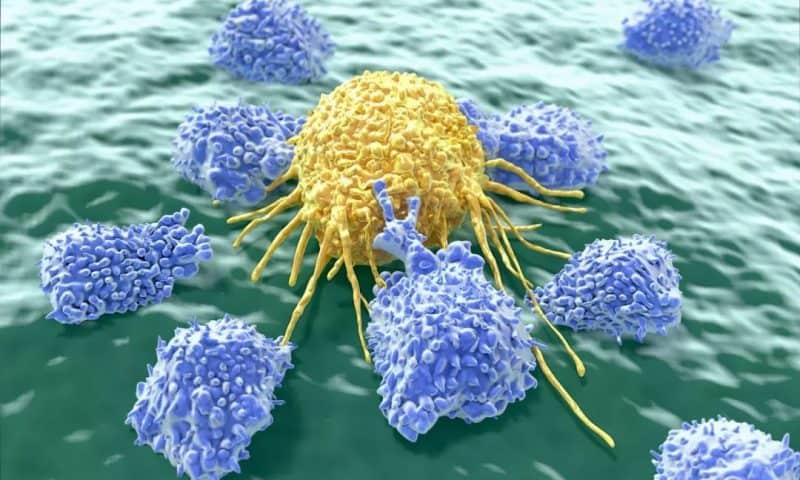
Clinical stage immuno-oncology innovator GT Biopharma, Inc. (NASDAQ: GTBP) is targeting multiple hematologic and solid tumor cancers with its first-in-class NK cell engager protein biologic technology platform: TriKE™. TriKE™ is designed to rescue the patient’s native natural killer cell (NK cell) population. Additionally, TriKE™ has the unique ability to be a self-sustaining,
self-activating, target-directed NK cell therapeutic agent – resulting in a persistent, 24/7 serial cancer cell killer, lasting for 60-90 days – without the need to transplant NK cells in patients or be co-administered with other therapeutic agents. For these reasons, GT Biopharma’s novel TriKE™ therapy will be significantly less expensive than other cancer treatments, opening the door to an off-the-shelf therapeutic for patients.
GT Biopharma recently reported updated interim Phase I/II clinical trial results for its lead therapeutic candidate, GTB-3550 TriKE™. GTB-3550 is currently being evaluated for the treatment of high-risk myelodysplastic syndromes (MDS) and refractory/relapsed acute myeloid leukemia (AML). Highlights of the results include: