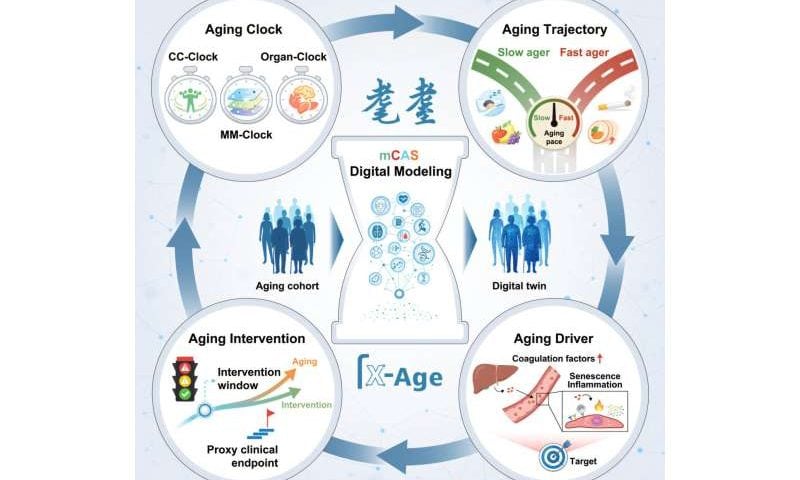
Aging is a complex process, and precisely measuring how the human body declines has long been a challenge. Two people of the same chronological age can have very different health trajectories. Scientists have also struggled to move beyond identifying aging markers to pinpointing what actually drives aging itself.
Now researchers from China’s Aging Biomarker Consortium (ABC) have built a computational framework—the Digital Aging Twin—to study aging at the individual level in order to predict biological age and track the different aging rates of individual organs.
The study, which was conducted by researchers from the Institute of Zoology, the China National Center for Bioinformation (Chinese Academy of Sciences), Xuanwu Hospital of Capital Medical University, and seven other institutions, marks a major breakthrough by moving from simply describing aging to systematically quantifying it, potentially paving the way for future interventions.
The findings were published in Cell on May 8.
Building a massive aging dataset
The team recruited 2,019 healthy individuals aged 18 to 91 from Chinese cities including Beijing, Quzhou, Ningbo, and Nanchang to create a standardized multicenter cohort called mCAS (multicentric Chinese Aging Standardized). The researchers collected data on 240 parameters for each participant, using clinical tests, cognitive and motor function assessments, brain and retinal imaging, gait analysis, and several layers of molecular data, including DNA methylation, RNA transcripts, proteins, metabolites, and gut microbiomes.
This dataset, comprising more than a billion high-quality data points, serves as the foundation for a three-tiered system of “clocks” to measure aging.
How the three-tier clock works
The first tier is the core capacity clock, which integrates 240 physiological indicators to reflect overall functional decline. The second and most powerful tier is the multimodal clock. It integrates multiple layers of molecular data (“omics” layers) via a deep learning process that employs “attention mechanisms”—which prioritize the most informative data—to quantify the contribution of different data types. This multimodal clock predicts chronological age with a mean absolute error of only 3.87 years, outperforming all single-omics clocks. The third tier comprises organ-specific clocks for the brain, liver, lungs, muscles, blood vessels, and skin, each based on clinical markers, plasma proteins, and imaging features.
One of the most striking findings is that organs age asynchronously. For example, the liver reaches a critical aging inflection point around age 40, whereas the brain’s aging accelerates at around age 50. The analysis also uncovered two major nonlinear waves of aging-related change: one occurring between ages 40 and 50, and another between 60 and 70.
Pinpointing coagulation factors as drivers
Seeking the causes of these aging-related changes, the researchers analyzed plasma proteomics, examined stained liver tissue from human donors, and conducted experiments using human cell cultures and animal models. They identified age-driven accumulation of liver-derived coagulation factors—particularly F13B, as well as F9 and F10—as a direct driver of vascular and systemic aging.
For example, when human aortic endothelial cells were exposed to these factors, they showed clear signs of senescence: elevated aging markers, impaired tube formation (a measure of blood vessel health), and increased inflammation. Similarly, injecting F13B into mice accelerated aging across multiple tissues, including the liver, heart, aorta, and kidney, accompanied by immune cell infiltration and inflammatory signals. These results show that coagulation factors are not just passive biomarkers but actionable drivers of aging.
Simplifying clocks for clinical use
To make the aging clock approach clinically practical, the researchers developed simplified “proxy clocks” using just 100 to 108 plasma proteins. These protein-based proxies closely match the predictions of the much more complex core capacity clock and organ clocks, suggesting that a relatively simple blood test might one day provide a comprehensive aging assessment.
The study also identified lifestyle factors influencing biological aging. Greater fruit intake, consistent sleep routines, and moderate walking were linked to slower aging. In contrast, smoking, insufficient sleep, and high meal frequency were associated with accelerated aging.
Implications for China’s X-Age Project
This research marks the first proof-of-concept achievement of the X-Age Project (also known as “耄耋”), a major national initiative led by the Aging Biomarker Consortium to build a comprehensive system of aging clocks for the Chinese population.
Although the current aging-clock framework is built on cross-sectional data, it is being continuously refined with longitudinal follow-up data and larger, more diverse populations. Future updates will also generate lower-cost and more sensitive detection approaches.
Despite the limitations of using cross-sectional data, the study has far-reaching implications for aging research as a whole. The Digital Aging Twin framework represents a fundamental shift in aging science—from description to prediction and from identifying correlations to pinpointing drivers.
Researchers now have a standardized, quantifiable, and interpretable system that can tell how fast a person is aging, which organs are aging most rapidly, and where interventions might be most effective.