Long-acting antiviral medications are transforming HIV prevention and care, requiring only minimalistic dosing. But as the use of lenacapavir expands, scientists are probing a critical question: If the virus evolves resistance, does it also pay a cost?
Lenacapavir is widely considered a trailblazing drug requiring only two injections annually. In 2024, it was hailed as Science magazine’s “Breakthrough of the Year.” Yet it isn’t the mere two doses that make the medication unique: it is the drug’s target. Lenacapavir zeroes in on the viral capsid, the outer protective shell that guards HIV’s genes. The drug was approved in the United States and Europe last year.
In a new study published in Science Translational Medicine, investigators have shown that HIV can acquire mutations that cause resistance to lenacapavir—but at a huge cost to the virus itself.
Mutations that are capable of promoting resistance to lenacapavir can also impair the ability of the virus to replicate. Indeed, the most lenacapavir-resistant viruses were also those with the poorest ability to replicate, researchers found.
“Lenacapavir is approved as a stand-alone twice-yearly option for HIV-1 preexposure prophylaxis in the U.S. and Europe and as a twice-yearly subcutaneous injectable in combination with other antiretrovirals for heavily treatment-experienced people with multidrug-resistant HIV-1,” Dr. Nina Pennetzdorfer, lead author of the research, writes in the journal.
It took 20 years to develop lenacapavir
The drug was developed at Gilead Sciences in Foster City, Calif., over a 20-year period, and unlike previous HIV antiviral medications, it is the first HIV drug to target the capsid instead of an enzyme.
The HIV capsid is a complex geometric structure called a fullerene cone that self-assembles from repeating protein units. It’s like a battlefield tank when it comes to breaking into cells and thwarting the most potent forces of the immune system.
“This capsid shell protects the replicating viral genome as it infects new cells and travels to the nucleus,” added Pennetzdorfer, an investigator at Gilead. “The arrangement and structure of the capsid protein are crucial for capsid stability and function during the viral infection cycle.”
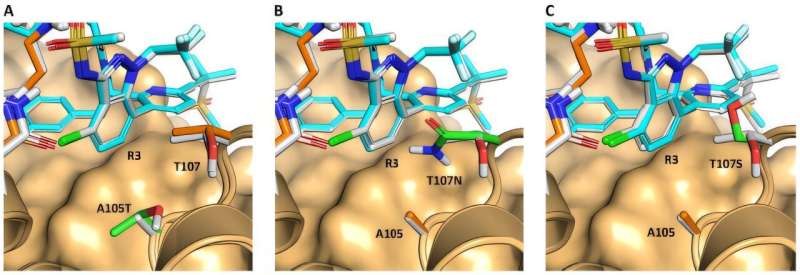
Pennetzdorfer and her team comprehensively defined 84 capsid mutants derived from either clinical trial isolates or site-directed mutants—viruses that scientists engineer in the lab then introduce a precise mutation in the capsid gene. The process is called site-directed mutagenesis.
The team then cloned these capsid mutations into new, replicating HIV clones. The studies revealed that the virus could evolve resistance to lenacapavir because some mutations rearranged the structure of the capsid’s binding pocket for the drug.
HIV resistant to lenacapavir pays a big price
Yet some of the most formidable resistance mutations came with a measurable fitness cost. Under standard laboratory conditions, those viral variants replicated less efficiently, suggesting that drug-resistant strains might struggle to compete with wild-type virus in the absence of lenacapavir.
In an analysis of Pennetzdorfer’s research, Drs. Manish Choudhary and Jonathan Li, both from Harvard Medical School and the division of infectious diseases at Brigham and Women’s Hospital in Boston, said the most important aspect of the study was unmasking potential resistance pathways.
“Three main lenacapavir resistance pathways emerged,” Choudhary and Li asserted, also writing in Science Translational Medicine.
“The first centered on M66I [a mutation], which had been the dominant pathway [to lenacapavir resistance] in heavily treatment-experienced participants in the CAPELLA clinical trial. Across assays, M66I alone resulted in large-fold changes in the median effective concentration value of lenacapavir, confirming it as a high-level resistance pathway.
“The second pathway involved Q67H and K70H/R [mutations], most commonly seen in the CALIBRATE trial,” they added, noting that the “third pathway included other resistance-associated mutations … which conferred low-to-medium resistance.”
Given the capsid’s central role in uncoating, nuclear import, and assembly, it’s not surprising, Choudhary and Li conclude, “that capsid mutations can come with substantial fitness costs.”
Pennetzdorfer and colleagues say insights from their study provide a foundation that may guide the design of next-generation capsid inhibitors. The study also provides a stronger guide for prescribing lenacapavir now.
“As [lenacapavir] moves into broader use for both PrEP and treatment, these findings highlight the importance of maintaining fully active companion drugs during HIV treatment, improving access to resistance testing and surveillance, and accelerating development of next-generation capsid inhibitors,” Pennetzdorfer concludes.