An epigenetic mechanism by which tumors manage to hide from the body’s immune defenses has just been described by an international scientific team led by the University of Liège and the Dana-Farber Cancer Institute. These findings open up avenues for new therapeutic strategies combining epigenetics and immunotherapy. The paper is published in the journal Cancer Cell.
Whole-genome doubling (WGD) is an event that frequently occurs during tumor development; it is observed in approximately 37% of primary solid tumors (tumors that form a physical, compact mass within an organ or tissue) and in up to 56% of metastatic tumors (secondary tumors that have developed after cancer cells have broken away from the primary tumor and migrated to other organs via the blood or lymphatic circulation). This duplication corresponds to a situation where the cancer cell ends up with twice the normal number of chromosomes. Long considered merely a marker of poor prognosis, this phenomenon was associated with resistance to treatment and increased genomic instability, without its mechanisms of action being clearly understood.
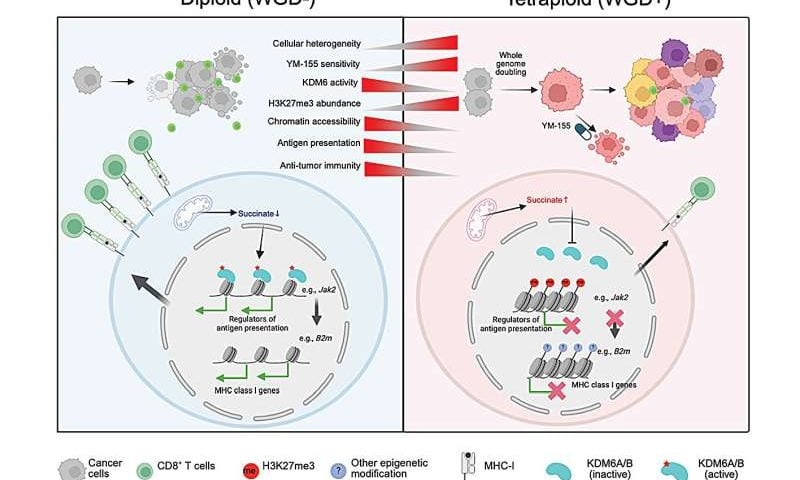
A new international study sheds significant light on how this mechanism develops, particularly in breast cancer. The researchers show that duplication does not merely disrupt the structure of the genome; it actively alters the relationship between the tumor and the immune system.
“Initially, genomic duplication increases the tumor cell’s visibility to the immune system,” explains Dr. Pierre Foidart, medical oncologist at the Liège University Hospital (CHU de Liège) and researcher at the University of Liège (fellowship of Foundation Against Cancer), and first author of the article. “But the cells quickly adapt by developing a strategy of invisibility.”
Normally, a cancer cell displays on its surface fragments of abnormal proteins called antigens. These antigens are presented by a structure known as the major histocompatibility complex class I (MHC-I), which acts like a “display window” allowing cytotoxic CD8+ T lymphocytes – immune cells specialized in destroying abnormal cells – to recognize and eliminate the cancer cell.
At the same time, certain cells of the innate immune system, particularly NK cells, produce a signaling molecule called interferon gamma (IFN-γ). This cytokine increases the amount of MHC-I present on the surface of cancer cells, thereby improving antigen presentation to CD8+ T lymphocytes.
Once activated, CD8+ T lymphocytes also produce interferon gamma, further enhancing the recognition of tumor cells. This creates a positive feedback loop that progressively amplifies the anti-tumor immune response.
“What our study demonstrates is that genome-doubled cells eventually switch off the genes responsible for MHC-I production, and therefore for antigen presentation,” continues Pierre Foidart. “They no longer display antigenic fragments on their surface and no longer respond correctly to interferon signals. T lymphocytes, deprived of the information they need to recognize the threat, pass by the tumor without launching an attack.” The cancer cell is still there, however, but it has become, in a sense, invisible to the immune system.
This camouflage is not genetic but epigenetic (the set of mechanisms that regulate genes without altering the DNA sequence). The DNA remains intact, but its expression is modulated by metabolic changes that lead to transcriptional repression of immune genes.
“This mechanism is driven in part by the PRC2 complex, an epigenetic regulator. And this is precisely what distinguishes epigenetic modifications from genetic mutations: epigenetic modifications are reversible, unlike genetic mutations,” says Dr. Kornélia Polyak, Professor of Medicine at Dana-Farber Cancer Institute, Harvard Medical School, and a co-leader of the Dana-Farber Harvard Cancer Center Cancer Cell Biology Program.
“By pharmacologically targeting this complex, it is possible to partially restore antigen presentation and make WGD-positive tumor cells recognizable to the immune system once again, while selectively inhibiting the growth of WGD-positive tumors.”
These findings have potential implications for clinical practice. Whole-genome duplication could eventually serve as a biomarker for stratifying patients and guiding treatment decisions, particularly towards combinations of epigenetic inhibitors and immunotherapy. Clinical studies will, however, be necessary to validate these approaches.
Pierre Foidart is currently continuing his research on this topic in collaboration with Professor Pierre Close (ULiège), with the aim of developing methods for detecting whole-genome duplication that are more accessible than whole-genome sequencing, which remains costly and is rarely used in routine clinical practice.