Lymphoma itself, not just treatment, can trigger rapid immune system and tissue aging
A new study led by a team of researchers at Moffitt Cancer Center reveals that lymphoma can accelerate the biological aging of the immune system and other tissues, providing new insight into how cancer reshapes the body beyond tumor growth.
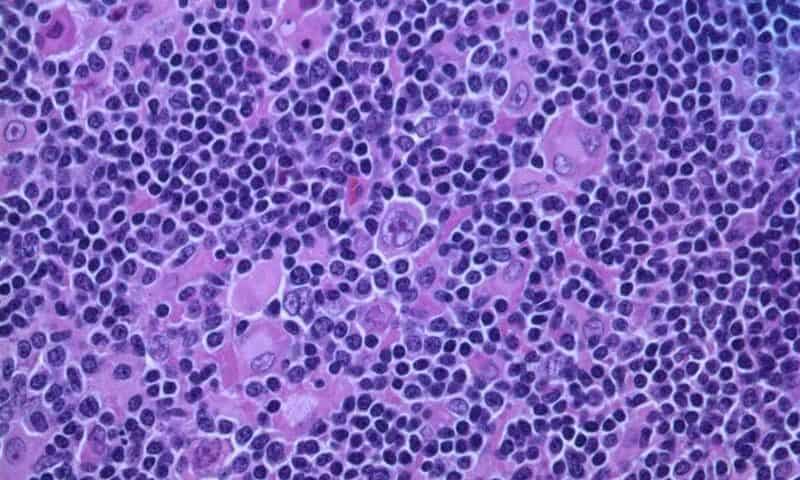
The study, published in Cancer Cell, shows that B cell lymphoma rapidly transforms young T cells, which are key immune fighters, into a state resembling those of T cells in much older individuals. These changes included increased inflammation, impaired protein balance and altered iron regulation. The effects were not limited to immune cells. Markers of aging also appeared in the blood vessels, kidneys and intestines.
“Cancer doesn’t just grow in isolation; it has widespread effects on patients. We found that lymphoma alone, without treatment, is enough to provoke systemic signs of aging,” said John Cleveland, Ph.D., senior author and chief scientific officer at Moffitt. “This helps explain why many cancer patients experience symptoms typically associated with aging.”
The findings challenge the long-held belief that accelerated aging in cancer patients is primarily caused by treatments like chemotherapy or radiation. While those therapies are known to age cells, this study shows that the cancer itself can push immune and tissue systems into an aged state.
“Our results also suggest there may be opportunities to reverse some cancer-driven aging effects,” said Rebecca Hesterberg, Ph.D., the study’s lead author and a researcher in Moffitt’s Department of Tumor Microenvironment and Metastasis. “By understanding the biology, we can begin to think about interventions that not only treat the cancer but also protect or even restore healthy immune function.”
Researchers discovered that lymphoma-exposed T cells accumulated excess iron, making them resistant to a type of cell death called ferroptosis. They also exhibited defects in protein quality control, a hallmark of aging. Some of these changes were reversible when tumors were eliminated in animal models, pointing to new therapeutic opportunities.
With the global population aging and cancer risk rising with age, the study underscores the importance of understanding how cancer interacts with aging biology.