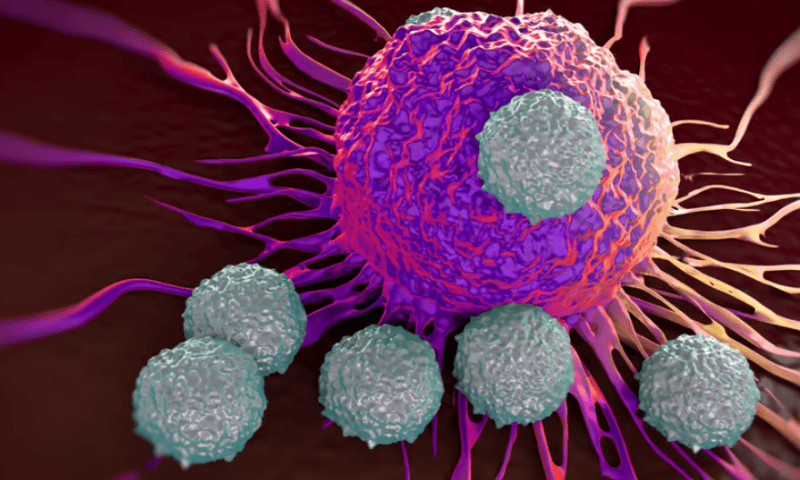
Verastem, Quanta and Frontier demonstrate KRAS inhibitors’ potential in preclinical data
Verastem Oncology, Frontier Medicines, Quanta Therapeutics and more shared preclinical updates at the American Association for Cancer Research’s (AACR’s) annual meeting in San Diego in early April, adding to the ever-growing list of companies vying to bring forth therapies against the once-“undruggable” target.