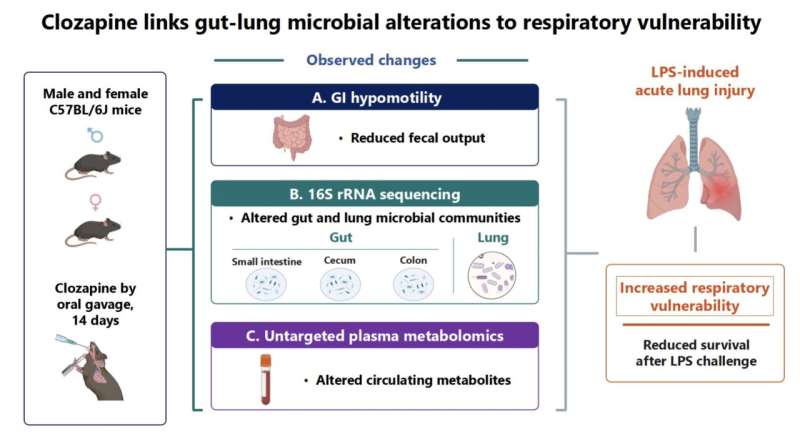
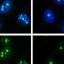
Why do cells age—and why do we lose our energy and vitality as we get older? This question is one of the central challenges of modern biomedicine. The focus is particularly on mitochondria—tiny cellular organelles long known as the cell’s powerhouses but now understood as dynamic control centers that not only produce energy, but also coordinate cellular communication, adaptation, and many of the processes essential for life.
They supply us with the energy that our body needs for movement, growth, and repair processes. But as we age, these powerhouses begin to slow down. It has long been known that their function declines with age. But until now, the mechanisms driving this gradual decline have been poorly understood.
Focus on membrane lipids
For a long time, it was assumed that genetic damage within the mitochondria themselves was primarily responsible. A study now published in Nature Communications by an international research team led by Dr. Maria Ermolaeva of the Leibniz Institute on Aging—Fritz Lipmann Institute (FLI) in Jena provides a surprising answer to this question: A key factor appears to be the imbalance in the structure of the mitochondrial network, which is caused by the absence of a major lipid in the membrane composition.
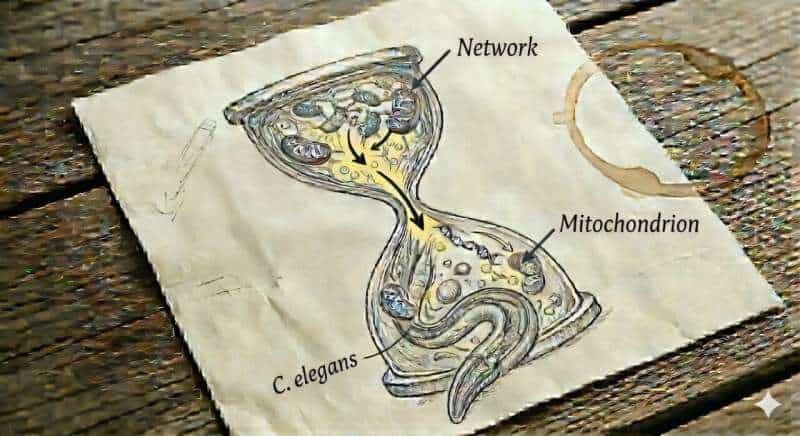
The focus is on phosphatidylcholine—a fundamental lipid that is a major component of biological membranes. It ensures that membranes remain flexible and can dynamically reorganize themselves. Precisely this property is crucial for so-called “mitochondrial fusion”—a process in which individual mitochondria merge into networks. These networks are necessary for cells to distribute key molecules—such as cellular energy equivalents, metabolic products, DNA, and signaling molecules—and facilitate their exchange, thereby preventing imbalances and replacing damaged components.
The study shows that the body’s production of phosphatidylcholine declines with age, leading to increased fragmentation and dysfunction of mitochondrial membranes. When genes involved in phosphatidylcholine synthesis were deactivated in young worms, their mitochondria in the cells quickly began to look “aged.”
The researchers were particularly fascinated by how closely these changes resembled the mitochondria typically observed in chronologically old organisms. Even more striking was the observation that the mitochondria regained a more youthful structure within just two days when the worms were fed phosphatidylcholine or its precursor, choline.
“We were surprised ourselves by how strongly this molecule influences the structure, connectivity, and function of mitochondria,” explains Dr. Tetiana Poliezhaieva, the study’s first author.
‘Butterfly effect’ of a small biochemical change
What initially sounds like a small biochemical change has far-reaching consequences (Butterfly effect). Normally, mitochondria form a dynamic network within the cell, which can constantly adapt to new demands. With age, however, this network becomes increasingly unstable. “You can imagine the whole system as a finely branched power grid that becomes increasingly damaged with age: connections break down and currents stall,” explains Dr. Ermolaeva, the study’s lead author.
“Although energy production continues, it becomes less efficient and sustainable, and energy can no longer be distributed flexibly.” As a result, cells gradually lose their “metabolic plasticity,” meaning their ability to quickly and efficiently adapt to changing energy demands.
This adaptability is essential for maintaining healthy function over time, not only at the level of individual cells but also across tissues and whole-body physiological systems. Its decline is therefore increasingly recognized as a key feature of aging, and it is also closely associated with diseases such as diabetes.
Methodological approach: From worms to humans
To decipher the underlying mechanisms, the research team combined multiple complementary model systems, including the nematode Caenorhabditis elegans, human cell cultures, and large-scale clinical patient data. Using a longitudinal, across-aging approach, they integrated extensive datasets covering proteomic and lipidomic profiles, genetic variation, gene expression, and metabolic activity in humans.
This multi-layered strategy allowed them to connect molecular changes observed in model organisms with patterns seen in human aging. This integrative approach—combined with experimental validation and whole-body functional analyses in worms—made it possible to uncover a direct mechanistic link between gradual molecular changes and systemic aging processes.
New insights into the aging process
The study found that, in addition to accumulating genetic damage, age-related changes in lipid synthesis also contribute to mitochondrial dysfunction. These findings expand our understanding of mitochondrial aging by identifying membrane lipid dynamics as an additional key factor.
A longitudinal comparative study of different life stages of the nematode was also of interest. The data suggests that aging does not proceed uniformly, but rather in phases with different biological breaking points. First, cells lose their capacity to cope with stress, alongside impairments in protein homeostasis—the system that maintains protein stability. This is followed by metabolic and, finally, epigenetic changes.
Sex-specific differences in lipid metabolism were also found: The strongest relative decrease in phosphatidylcholine levels was detected in human metabolome data in women around the age of menopause. “This observation is particularly noteworthy, as it coincides with a time when many women report a significant decline in energy levels and the onset of persistent fatigue,” adds Dr. Ermolaeva.
Aging biology can be modulated
Perhaps the most important finding of the study, however, lies in the reversibility of aging-associated failures: through a targeted increase in phosphatidylcholine levels—for example, via diet—the mitochondrial networks in old C. elegans stabilized, and the cells began producing energy more efficiently again. This indicates that at least some aspects of aging can be substantially restrained, allowing for a longer period of healthy life—and that targeted interventions in metabolism could make a difference.
“Our work shows that both mitochondrial aging and broader systemic aging are, at least in part, modifiable. If we understand the underlying processes, we may be able to take targeted countermeasures,” summarizes Dr. Ermolaeva. Whether and how these findings can be translated into concrete therapies for humans must be clarified in further studies. The role of nutrition is particularly interesting in this context: certain nutrient supplements might help stabilize cell function in old age.
Finally, this study shows that phosphatidylcholine supplementation can serve as an effective anti-aging intervention even when initiated at middle or advanced age. Overall, the study provides an important impetus for aging research. It shifts the focus from irreversible decline to modifiable processes, thereby offering hope that healthy aging can be more actively shaped in the future.