Autism spectrum disorder (ASD) is a neurodevelopmental condition estimated to affect approximately 1 in 100 children worldwide. This condition is characterized by differences in how people communicate and interact with others, as well as restricted interests and repetitive behaviors.
Some autistic people have been found to exhibit mutations in SHANK3, a gene that encodes a key protein contributing to the formation and maintenance of junctions between nerve cells (i.e., synapses). Past studies have shown that approximately 40% of autistic individuals with SHANK 3 haploinsufficiency (i.e., presenting one functional copy of the gene instead of 2) tend to lose previously acquired social, communication-related or motor skills over time.
Researchers at Yale University School of Medicine recently set out to explore the possible contribution of neuroinflammation to the behavioral regression observed in many cases of SHANK3-associated ASD. The findings of their study, which focused on a mouse model of SHANK3-related autism, are published in Molecular Psychiatry.
“We previously reported that significant behavioral regression in a small cohort of patients with SHANK3 haploinsufficiency, triggered by subclinical infections, responded to immunomodulator treatments,” wrote Sheng-Nan Qiao, Sung Eun Wang and their colleagues in their paper. “We hypothesize that behavioral regression results from the interplay between SHANK3 deficiency and neuroinflammation.”
Studying a mouse model of ASD
Qiao, Wang and their collaborators performed a series of experiments involving mice that exhibited a partial loss of the SHANK3 gene as well as regular mice. As part of these experiments, they injected the mice with lipopolysaccharides (LPS), a molecule that triggers immune responses.
The team then observed the mice’s behavior following LPS injection, comparing it to that of regular wild mice. Interestingly, they found that injecting LPS and triggering immune responses altered the behavior of SHANK3-deficient mice over time, with the animals exhibiting anxiety-like behaviors, repetitive actions and difficulties with movement.
“Using Shank3 exon 4–22 deletion heterozygous mutant (Sh3+/−) mouse, which shows no significant behavior impairments, we established a preclinical model—Shank3 haploinsufficiency mouse undergoing a systemic inflammation challenge via intraperitoneal injection of LPS,” wrote the authors.
“We found that, two weeks after LPS challenge, wild-type mice (WT) recovered but Sh3+/− mice exhibited motor impairment, anxiety-like behaviors, and excessive grooming, similar to Shank3 exon 4–22 deletion homozygous mutants. Anti-inflammatory treatment partially reversed LPS-induced behavioral changes.”
The researchers found that anti-inflammatory treatments reversed the behavioral changes observed in the mice after LPS injection, reducing repetitive grooming, anxiety-like behaviors and improving their motor skills. These results suggest that neuroinflammation contributes to the behavioral regression observed in the SHANK 3-deficient model of ASD, and thus, it could be a promising therapeutic target.
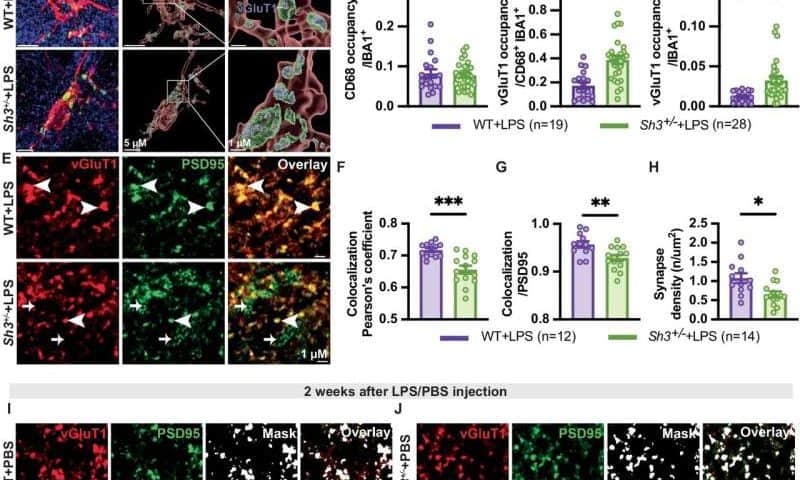
“Transcriptomic analysis revealed upregulation of neuroinflammation-related genes and downregulation of synaptic function-related genes in LPS-challenged Sh3+/− mice,” wrote Qiao, Wang and their colleagues. “Especially, pro-inflammatory genes and microglia markers were overly activated that may result from the increased toll-like receptor 4 (TLR4) in Sh3+/− mice. Microglia overactivation elevated synapse engulfment and disrupted synaptic protein may underlie LPS-triggered worsened behavior phenotypes in Sh3+/− mice.”
The link between genetic vulnerability and immune activation
This recent study could soon inspire other researchers to further explore the link between neuroinflammation and behavioral regression in ASD linked with SHANK3 haploinsufficiency.
If the team’s findings are validated in humans, they could also pave the way for new therapeutic strategies tailored for autistic people with this specific genetic vulnerability.
By reducing the immune system’s activation, these new treatments might prevent or reverse behavioral changes that interfere with the daily functioning of individuals with SHANK3 haploinsufficiency-related ASD.
“Together, our findings indicate that neuroinflammation increases the penetrance of behavioral impairment in Shank3 haploinsufficiency mice and supports a potential mechanism for behavioral regression in human SHANK3 related disorders for future investigations,” wrote the authors.